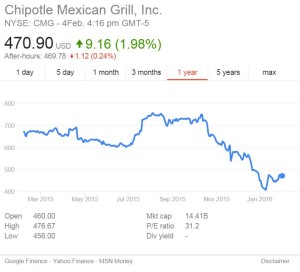
The Senate’s (mostly unfunded) agenda for the FDA
Food Chemical News reports that the Senate Appropriations bill comes with report language instructing the FDA to:
- Conduct a risk assessment to see how harmful it really is to eat Listeria-tainted frozen vegetables [these, presumably, would be cooked before eating]. CRF Frozen Foods had to recall of 358 organic products after 7 people became ill.
- Release no sodium guidelines before the National Academies of Science has a chance to update the Daily Reference Intake (DRI) for sodium [which will take years]. The report says “it is “imperative that any guidance be issued using the latest sound science…based upon an updated DRI report.” House directives said the same thing. [The FDA has just released voluntary sodium guidelines for public comment].
- Release its overdue report on food traceability systems: “The Committee directs the FDA to collaborate with science-based international and industry-led food traceability initiatives of the type recommended by the pilot projects…[and] to make publicly available information on FDA’s efforts to encourage…traceability initiatives.”
- Reconsider its E. coli standard. The Senate views this standard as too stringent and disruptive of raw milk cheese businesses.
- Actively engage stakeholders in best practice standards for medical foods. “They should work together on “forming a framework for a distinct regulatory pathway for medical foods that does not encumber its progress towards approval for patient use.”
- Better enforce standards for antibiotics in shrimp imports.
- Sample olive oil bottles at retail to determine if they are adulterated, and report findings to Congress.
- Report on efforts to implement regulations for the growing commercial human milk industry.
Some of these directives would improve food safety, but some would not.
All make more work for the FDA.
Does the Senate also plan to appropriate sufficient funds for the FDA to do all this in addition to what it is already doing? Want to take bets?