Pet Food I. Is the FDA doing enough to ensure its safety?
Two items about pet food this week. Today: safety. Tomorrow: environmental sustainability.
I have a long-standing interest in pet food, which I view as an integral—essential—component of our overall food system , not least because pet food uses food components that would otherwise be wasted. I co-authored a book about pet food issues: Feed Your Pet Right.
If there are problems with the safety of pet food, you can bet those problems will occur in the food supply for humans. I wrote a book about that too: Pet Food Politics: The Canary in the Coal Mine.
I am not the only one interested in such topics. Phyllis Entis has written two books crticizing the safety hazards of pet foods.
- TAINTED. From Farm Gate to Dinner Plate, Fifty Years of Food Safety Failures
- TOXIC. From Factory to Food Bowl, Pet Food Is a Risky Business
She keeps me up-to-date on the latest problems that arise.
I don’t know how closely you’re watching the pet food industry these days, but I thought this piece I just posted might interest you: When ‘truths’ collide: Darwin’s, ANSWERS, and the FDA
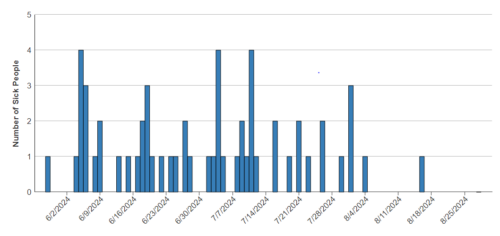
This was an account of an FDA Advisory and its non-consequences: Do Not Feed Certain Lots of Darwin’s Natural Selections Pet Food Due to Salmonella and Listeria monocytogenes.
The U.S. Food and Drug Administration is cautioning pet owners that FDA samples of five Darwin’s Natural Pet Products raw cat and dog food made by Arrow Reliance, Inc. tested positive for Salmonella and a sixth FDA sample tested positive for Salmonella and Listeria monocytogenes (L. mono)…If you have any of the Darwin’s Natural Selections pet food product listed above, stop feeding the product to your pets and throw it away in a secure container where other animals, including wildlife, cannot access it. Do not donate the food.
The bigger story here, as Phillis Entis explains, is that the FDA did not force the company to recall the products; it asked Darwin to rissue a recall volunarily.
But in this case, the company resisted. It voluntarily decided not to do the recall. Pet Food Industry (an essential source of information) tells this story.
In a September 20, 2024, memo from the company to consumers which was sent to Petfood Industry, Darwin’s Natural Pet Products had this to say given the FDA’s public notice regarding the affected lots of cat and dog food.
The memo is worth a look. It says things like this:
- At Darwin’s, the health and safety of your pets is our absolute top priority, and we take rigorous steps to support their well-being and to foster strong lines of communication with you and our fellow community members.
- It is also very important to know that the FDA has received no consumer complaints regarding any of these lots.
- We find the FDA’s public notice to be wholly unnecessary, and ultimately, based on flawed regulatory decision-making, and we have taken steps to make this position clear to the agency.
Oh.
Food safety lawyer Bill Marler asks: Will the FDA use its Recall Authority to Mandate a Pet Food Recall due to Salmonella and Listeria? .The FDA has that authority. It did not use it.
That leaves you on your own to make sure you do not buy Darwin products for your pet.
The FDA provides plenty of information as a basis for you taking responsibility for such things.
Its advisory asks this question: Why is the FDA concerned about Salmonella and L. mono in pet food?
Pet foods contaminated with Salmonella and L. mono are of particular public health importance because they can affect both human and animal health. Pets can get sick from Salmonella and may also be carriers of the bacteria and pass it on to their human companions without appearing to be ill…The Federal Food, Drug, and Cosmetic Act requires that all animal foods, like human foods, be safe to eat, produced under sanitary conditions, contain no harmful substances, and be truthfully labeled. Refrigeration or freezing does not kill Salmonella or L. mono.
It provides these resources:
- Get the Facts about Salmonella
- Salmonella (Salmonellosis)
- Get the Facts about Listeria
- Listeria (Listeriosis)
Do we need more forceful regulation of pet food? Of course we do.