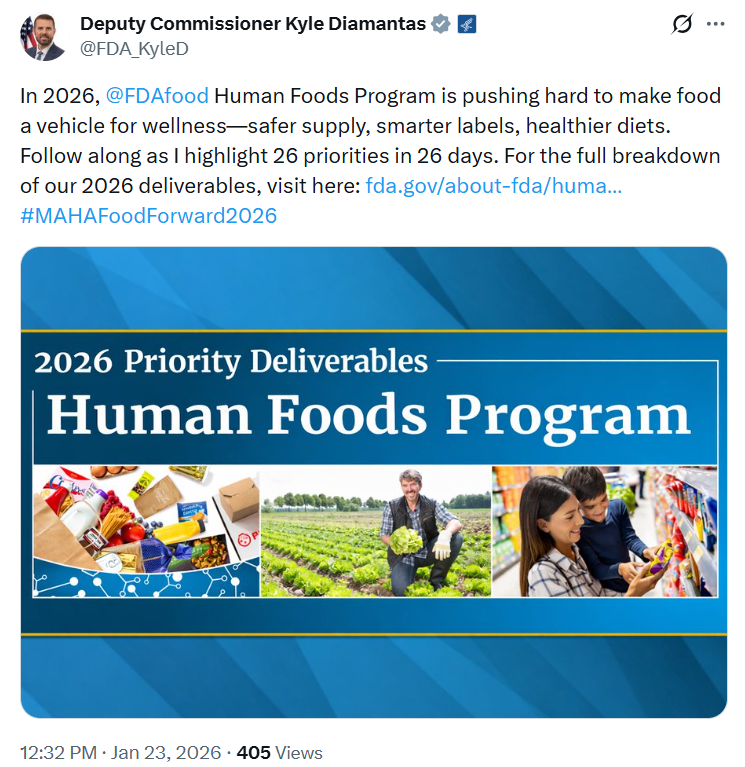
I thought this post on X was well worth a follow up. I went right to the site: Human Foods Program 2026 Priority Deliverables.
Its vision: “to ensure that food serves as a vehicle for wellness.”
Its mission: “to protect and promote the health and wellness of the American public through science-based approaches to prevent foodborne illness, reduce diet-related chronic disease, and ensure chemicals in food are safe.”
Its 2026 Priority Deliverables: these are listed in three categories: food chemical safety, nutrition, and microbiological food safety.
I. Food Chemical Safety (my selection and summary)
- Reform GRAS; regulate
- Review safety of food chemcials
- Conduct research on microplastics
- Establish action levels for cadmium and inorganic arsenic in baby foods
- Research consumer exposure to PFAS and other chemicals
- Regulate new dietary ingredients
- Modernize oversight of supplements
- Collect opinions on allergens; develop regulations
II. Reducing chronic disease through better nutrition
- Research ultra-processed foods; develop definition
- Research infant formula nutrient requirements
- Recruit experts to develop a UPF policy agenda
- Work toward issuing a front-of-package label to encourage healthier consumer choice and reformulation
- Implement the “healthy” front-of-package label
- Develop strategy to reduce added sugars
- Evaluate phase I targets for sodium reduction
- Issue guidance on food labeling for online shopping
III. Microbiological food safety
- Get states to take action
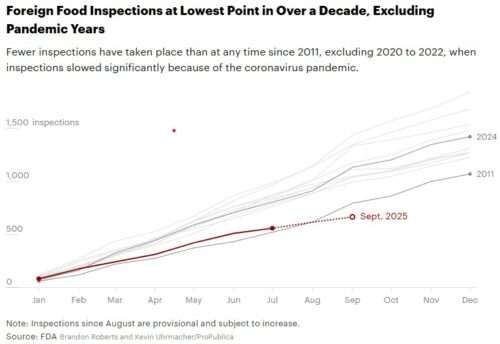
- Increase oversight of imported food
- Increase oversight of imported shrimp
- Train growers to prevent produce contamination
- Improve recall communication
Comment
I haven’t listed everything but this should give you the idea. Lots of this involves “research,” “develop,” and “evaluate.” Hardly any involves real regulation (except for chemical food additives). Still, this is, or could be, an impressive list.
The most pressing area is microbial food safety, because we are still seeing so many people made ill by contaminated food, especially infant formula.
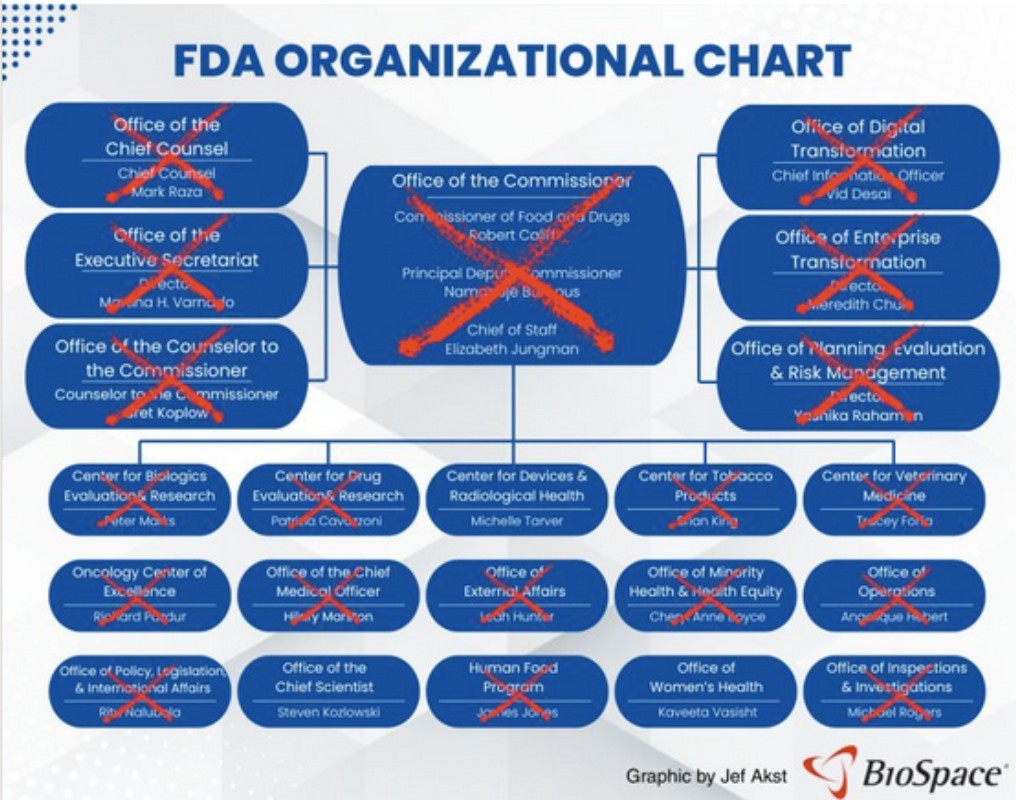
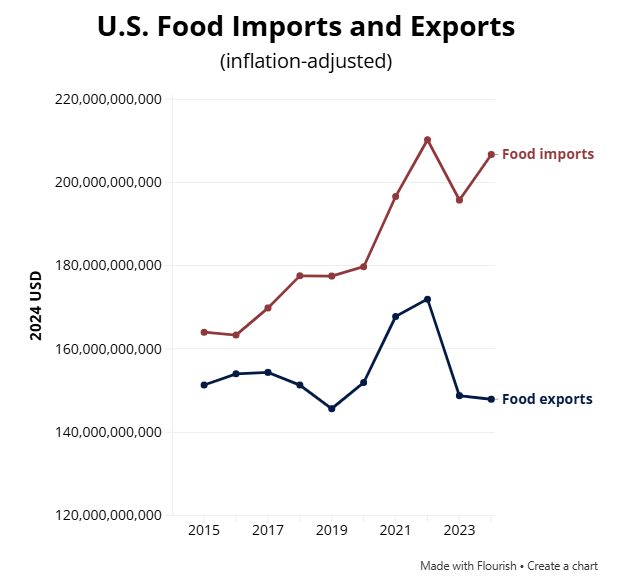
The big question: Where is the FDA going to get the resources needed to carry out this agenda? The FDA, already working for decades on an increasingly bare-bones budget for all it is required to do, eliminated a fifth of its workforce last year.
And this administration prefers personal responsibility as the primary approach to dietary health.
It would be great if the FDA could do all this in 2026. We are already in February. It has best get busy.