The FDA is an agency within the Department of Health and Human Services, now headed by Robert F. Kennedy, Jr. It is getting busy on carrying out Secretary Kennedy’s stated agenda. It took four actions of interest last week. Check out #3; it requires action.
I. Approved Three Food Colors from Natural Sources
Since the HHS and FDA announcement last month during a press conference at HHS on petroleum-based food dyes, more U.S. food manufacturers have committed to removing them within the FDA’s set time frame of the end of next year.
“On April 22, I said the FDA would soon approve several new color additives and would accelerate our review of others. I’m pleased to report that promises made, have been promises kept,” said FDA Commissioner Martin A. Makary, M.D., M.P.H. “FDA staff have been moving quickly to expedite the publication of these decisions, underscoring our serious intent to transition away from petroleum-based dyes in the food supply and provide new colors from natural sources.”
FDA approved color additive petitions for:
- Galdieria extract blue, a blue color derived from the unicellular red algae Galdieria sulphuraria (by petition from Fermentalg).
- Butterfly pea flower extract, a blue color that can be used to achieve a range of shades including bright blues, intense purple, and natural greens (Sensient Colors LLC)
- Calcium phosphate, a white color approved for use in ready-to-eat chicken products, white candy melts, doughnut sugar, and sugar for coated candies (Innophos Inc).
II. Announced top priorities for the Human Foods Program
FoodNavigator-USA report that Mark Hartman, who directs the new Office of Food Chemical Safety, Dietary Supplements, and Innovation, says the FDA soon will:
- Reveal how it will deal with the safety of chemicals in the food supply
- Create a new Office of Post Market Review to conduct risk reviews of chemical additives
- Increase transparency and stakeholder engagement in the review process
- Work through 70,000 comments on the FDA’s proposal for reviewing the safety of chemical additives
- Partner with the NIH to research how food additives affect children’s health
- Work with the food industry to phase out synthetic color additives
- Work through comments on sodium guidance
- Think about ways of addressing added sugars
- Identify ultra-processed foods as an “area of emerging study”
III. Extended the comment period for front-of-package labeling until July 15
We are taking this action in response to requests to extend the comment period to allow interested parties additional time to submit comments. Comments should be submitted to Regulations.gov and identified with the docket number FDA-2024-N-2910.
Recall: This is what the Biden FDA proposed. Here’s what I said about it (basically, we need something better).

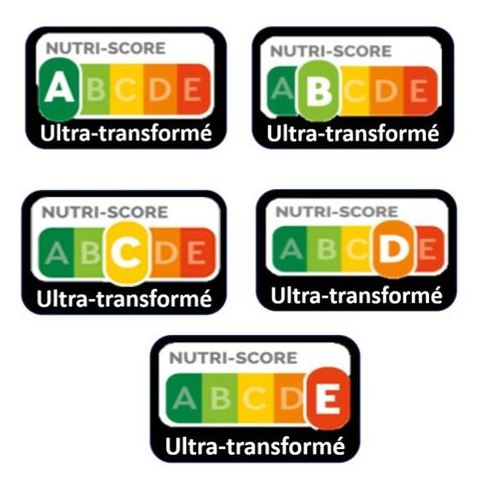
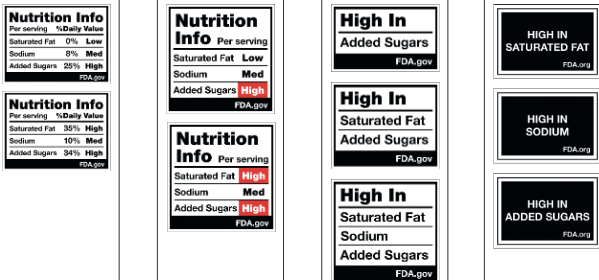
Here’s a real opportunity. If you want a front-of-package warning label like those in Latin America, here’s your chance to weigh in.
RFK Jr says he wants to Make America Healthy Again. One way to do that is to discourage sales of food products high in saturated fat, sodium, and added sugars, but also discourage sales of ultra-processed foods. Identifying foods as ultra-processed, on the basis of their chemical additives as well as their fat, sugar, and salt, would be an excellent step forward.
If you like the warning labels used in Latin American countries, send a note to the FDA Docket. You have until July 15 to do that.
IV. Announced a joint research initiative with NIH to address, among other unspecified questions,
- How and why can ultra-processed foods harm people’s health?
- How might certain food additives affect metabolic health and possibly contribute to chronic disease?
- What is the role of maternal and infant dietary exposures on health outcomes across the lifespan, including autoimmune diseases?
Comment
OK. This represents action or proposed action. My question: What will the FDA actually do? I’m particularly interested in the joint NIH research initiative on ultra-processed foods. Will NIH reverse its stance on Kevin Hall, whose research aimed to answer precisely that question? I will be watching all this with much curiosity.