USDA is closing buildings, relocating staff, and downsizing—a lot.
When President Trump was elected, he promised to downsize government. He is doing that, for better or worse. The latest move affects the USDA: GSA and USDA Unlock $1.6 Billion in Savings for Americans with Ag South Disposition.
Translation: The USDA is getting rid of the enormous, aging building it occupies across the street from its headquarters near the National Mall.
I love the positive spin: “The Ag South disposition will deliver significant value to taxpayers while advancing the Trump Administration’s objectives to reinvigorate, consolidate, and better utilize the federal real estate portfolio.”
Another account of this action reveals that two properties are involved, one of them currently housing the Food and Nutrition Service (FNS), the agency that runs food assistance programs.

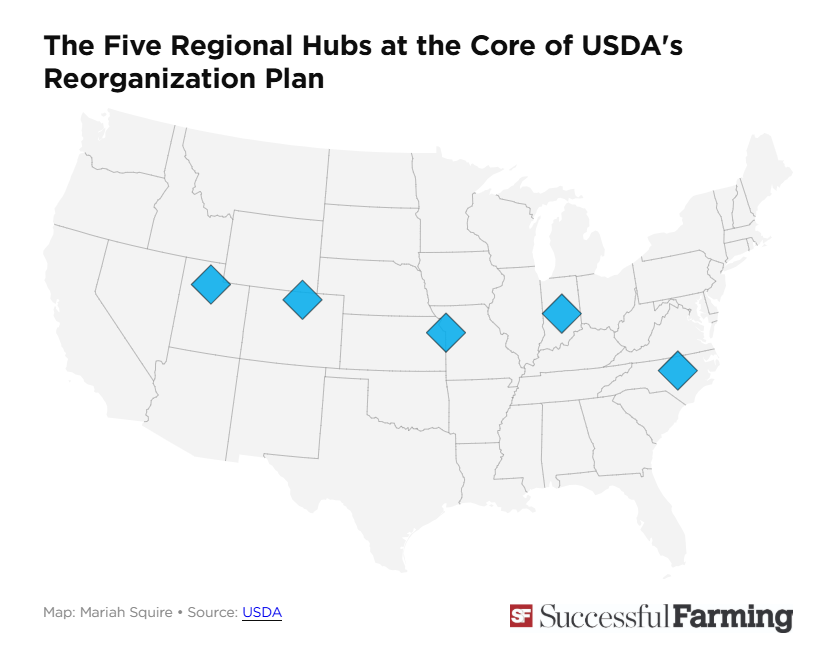
The USDA announced this plant last summer as part of the department’s major reorganization plan. Its point: to relocate staff to new USDA hubs.
As explained in yet another account, this is about relocating staff:
Thousands of USDA employees have taken buyouts and left the department over the past year amid Republican U.S. President Donald Trump‘s effort to reshape and reduce the size and footprint of the federal government. The USDA has said it is planning to relocate much of its remaining staff in the U.S. capital to hubs in North Carolina, Missouri, Indiana, Colorado and Utah.
From Government Executive, we learn:
The department announced the disposal of the South Building, which Rollins and other officials repeatedly described as dilapidated and mostly empty, last year as part of a larger reorganization that will push 2,600 employees out of the national capital region…USDA currently has 4,600 employees in the Washington area and is looking to shrink that number to 2,000…The department has already shed more than 15,000 employees from its initiative that allowed employees to sit on paid leave for several months before resigning.
Government Executive explains why this concerns me so much:
During Trump’s first term in 2019, the department relocated its Economic Research Service and National Institute of Food and Agriculture to Kansas City, over the objections of employees and some lawmakers. Following the move, both agencies lost more than half of their staff, leading to a significant loss of productivity from which it took the agencies years to recover. Under President Joe Biden, both agencies moved their headquarters back to Washington while maintaining their Kansas City offices.
…USDA solicited feedback on its reorganization plan last year, leading to 14,000 unique responses. Of those, 82% expressed negativity toward the plan, while 5% took a positive tone. Employees, lawmakers and stakeholders submitting the comments warned of a significant brain drain and disruptions to key farmer-support programs if the changes were implemented.
Oh no, not again.
I long considered the Economic Research Service to be the best kept secret in Washington, DC. It produced reliable, credible analyses of food issues. I considered it a national treasure and was devastated by its destruction (I wrote about this often on this site). In my view, the agency has never recovered from its loss of national experts. USDA says the FNS will remain in the DC area.
It’s hard not to see this as yet another attempt to undermine food assistance programs and make it harder for people to enroll in them.







