Weekend reading: Eric Schlosser on our cartel food system
In the Atlantic (to which I subscribe), Eric Schlosser writes: Do We Really Want a Food Cartel?
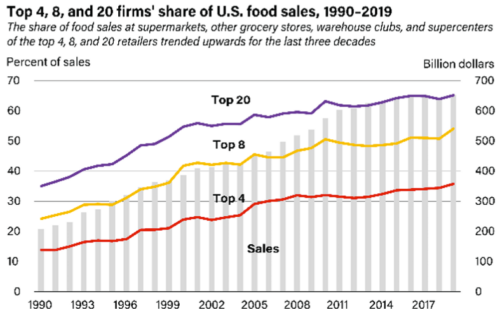
Mergers and acquisitions have created food oligopolies that are inefficient, barely regulated, unfair, and even dangerous.
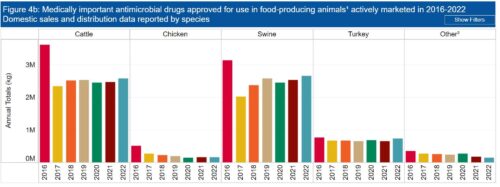
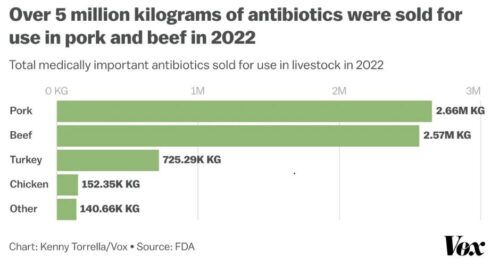
He begins with the Federal Trade Commission’s report on what happened to the US food supply during the COVID-19 pandemic: Feeding America in a Time of Crisis: FTC Staff Report on The United States Grocery Supply Chain and the COVID-19 Pandemic. This report focuses on the hazards of corporate consolidation for workers and consumers.
We see the effects in the grocery store. Prices are up and executive compensation even more so.
As Schlosser puts it,
When four companies gain a combined market share that is greater than 40 percent, an oligopoly has formed. The prices offered to suppliers, the prices charged to consumers, and the wages paid to workers are no longer determined mainly by market forces. Further, the power these corporations exert within their industries and the economy as a whole leads to a well-documented dynamic: Effective government regulation becomes difficult, whether because state and federal agencies are “captive” or because they are outmatched in terms of resources and personnel.
This brings us back to my post this week about the FDA. Schlosser describes the agency as “Poorly managed and notoriously reluctant to confront major food companies,” as shown by its inability to get on top of infant formula shortages.
Read his piece and see if he makes the case for his conclusion:
the fight against oligopolies and monopolies won’t be easy. A small number of corporations have tremendous political influence, expensive attorneys, and great skill at rigging markets in ways the public just can’t see.
We all need to pay close attention.